Imagine this: you are minding your own business, soaking up the sun, when suddenly a six-ton elephant strolls over, wraps its trunk around you, and picks you up. It’s a bummer way to start your Monday, especially considering that just yesterday you had a dik-dik nibbling on your left arm. Alas, this is the norm (though not always the end) for most of our leafy friends in the African savanna, where it’s not an easy place to be a tree.
Savannas are ecosystems co-dominated by trees and grasses, but the balance between these two functional groups varies widely across the African continent. This variability has large consequences for ecosystem structure and function. Tree cover, for example, is correlated with herbaceous diversity, aboveground carbon storage, and predator-prey dynamics. A central goal of savanna ecologists is to understand where, how, and why tree cover changes.
Predicting changes in vegetation structure is extremely difficult because savanna tree cover is determined by complex interactions between rainfall, fire, and herbivory that limit tree establishment and growth. The result is often a grassy landscape, sprinkled with short-statured trees that are perpetually vulnerable to disturbance, like herbivory. Understanding how trees cope with these interacting disturbances is key to explaining species distributions and competitive outcomes across environmental gradients.
In June and July 2025, I traveled to Mpala Research Centre (MRC) in central Kenya to investigate (1) how herbivory and rainfall affect savanna tree physiology and (2) how physiological strategies differentiate closely related, but highly dominantAcacia species. Our goal is to link species-specific physiological strategies to changes in tree growth and abundance across rainfall and herbivory gradients. To do this, we quantified leaf-level carbon gain and water stress in and outside of herbivore exclosures for the three most dominant Acacia species at Mpala Research Centre (Fig. 1).

Figure 1. (a) a heavily browsed Acacia mellifera and (b) Acacia brevispica tree. Both are consumed by herbivores but A. brevispica is heavily targeted due to its large leaves and small spines. Photo credit: Noah Hanisch. (c) vegetation in and outside of an herbivore exclosure. Photo credit: Evan Foster.
Research Site and Experimental Herbivore Exclosure
Mpala Research Centre is a 20,000-hectare thorn-scrub savanna and former ranch home to a diversity of wild and domestic herbivores, including elephants, giraffes, zebras, cattle, and camels. The site also hosts several long-term herbivore exclusion experiments, including the Ungulate Herbivory Under Rainfall Uncertainty (UHURU) experiment (Goheen et al. 2018). The UHURU plots were established in 2008 by Drs. Rob Pringle (Princeton University), Jake Goheen (Iowa State University), and Todd Palmer (University of Florida). The experiment is composed of 1-ha size-selective herbivore exclusion and control plots replicated across a 22-km north-south rainfall gradient (mean annual rainfall 450 – 650 mm). We focused our sampling within the control and total herbivore exclusion plots in the north and south of the research site (Fig. 2).
We chose to study the three dominant Acacia species at the site: A. brevispica, A. etbaica, and A. mellifera. Each of these species differs in their growth form, defense investment, and preference by herbivores. For example, A. brevispica is a species with large leaves and small spines and was found in 100% of the elephant diets and even several grazer diets sampled at MRC. In contrast, A. mellifera, a species with smaller leaves and sharp, hooked spines, was found in 59% of elephant diets (Kartzinel et al. 2015).
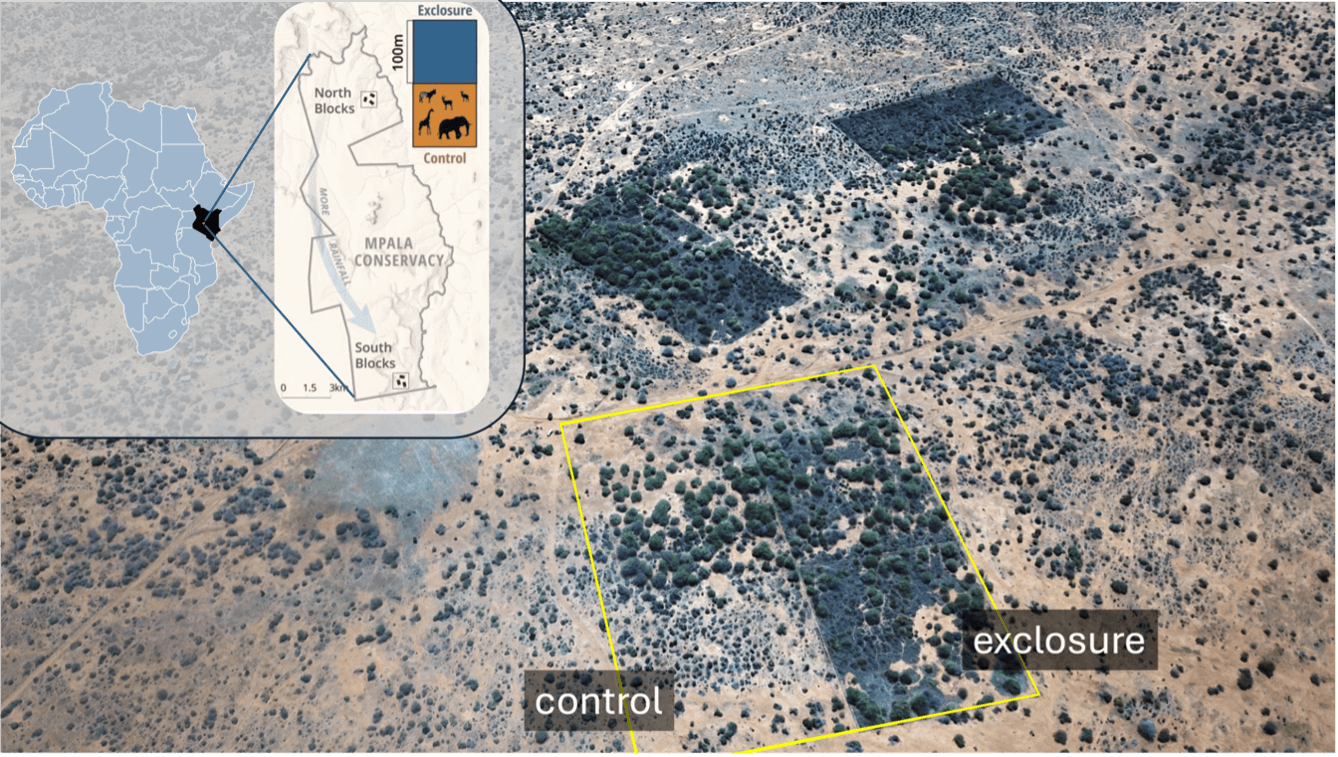
Figure 2. Mpala Research Centre in Laikipia county, Kenya. The site map shows a subset of the UHURU herbivory exclosures along the rainfall gradient. A treatment block is highlighted in yellow and is comprised of 4 herbivore treatments: control (bottom left), total exclusion (bottom right), megaherbivore exclusion (top left), and mesoherbivore exclusion that excludes everything larger than a dik-dik (top right). We sampled within the control plots and the total exclusion plots. Two other treatment blocks are in the background.
Using the LI-6800
We used the LI-COR LI-6800 Portable Photosynthesis System to measure instantaneous gas exchange and fluorometry for the three focal tree species, in and outside of herbivore exclosures in the north and south UHURU plots. We coupled these measurements with predawn (which was quite an adventure with lions roaring and elephants rumbling around us!) and midday leaf water potential to gain a better understanding of how stomatal conductance and assimilation rates respond to changes in water stress.
In addition to survey measurements, we conducted over 100 A-Ci curves, using the dynamic assimilation response technique (DAT), to understand differences in photosynthetic capacity and efficiency among the tree species and across the different locations.

Figure 3. (a) Practicing A-Ci curves. Don’t worry about the clouds, this was just for practice! (b) Measuring gas exchange on Acacia etbaica with the 2 cm2 fluorometer chamber head.
Along the way, we faced a few challenges. Fieldwork at MRC requires teams of researchers, research assistants, and askaris to help guide help guide the way and keep an eye out for wild animals. Additionally, Acacia trees are difficult to measure because they have very delicate leaves made up of dozens of tiny leaflets, which required us to correct for leaf area. The 2 cm2 chamber helped us overcome that challenge by making it possible to (nearly) fill the gasket. In addition, savanna trees ramp up and shut down their peak assimilation rates early in the day due to the intense heat and radiation during the afternoons, allowing us to start gas exchange measurements as early as 8:00 am – a first for me!
Next Steps
Our initial findings reveal physiological differences among the dominant Acacia species. Most notably, A. brevispica's water-spending strategy is more pronounced than the other species and maintains positive assimilation rates at remarkably low water potentials (-6 to -7 MPa). In the drier areas of the site, we found that A. brevispica also had higher Jmax, Vcmax, and maximum assimilation rates than the other species (Fig. 4), likely a result of having greater stomatal conductance under dry conditions. Our next steps are to connect these physiological strategies with the long-term growth data from the UHURU project (Alston et al. 2022) to better understand how species-specific strategies can drive community responses to herbivore exclusion and variation in rainfall.
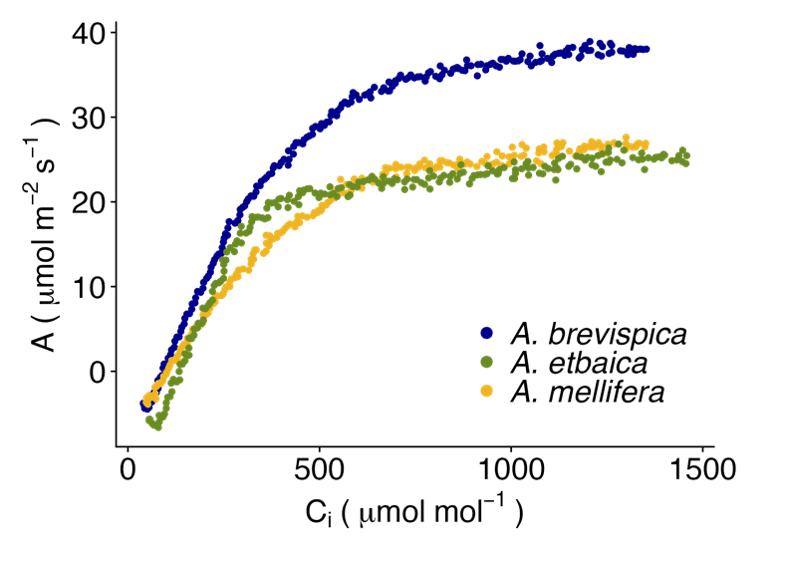
Figure 4. A-Ci curves for each of the focal Acacia species measured using the dynamic assimilation response curve method on the LI-6800. A. brevispicatends to have greater stomatal conductance than the other species and reach greater assimilation rates under high [CO2].

Emily Wedel is a postdoctoral researcher at the University of Notre Dame working with Dr. Tyler Coverdale. Her research focuses on understanding how the physiological strategies of dominant savanna plants promote their coexistence and scale-up to shift community- and ecosystem-level processes under environmental change.