Using Custom Chambers
Printable PDF: Using Custom Chambers
(6800_Note_Custom_Chamber_16768.pdf)
Download this content as a pdf that can be saved to your computer or printed.
Flow path and the chamber interface
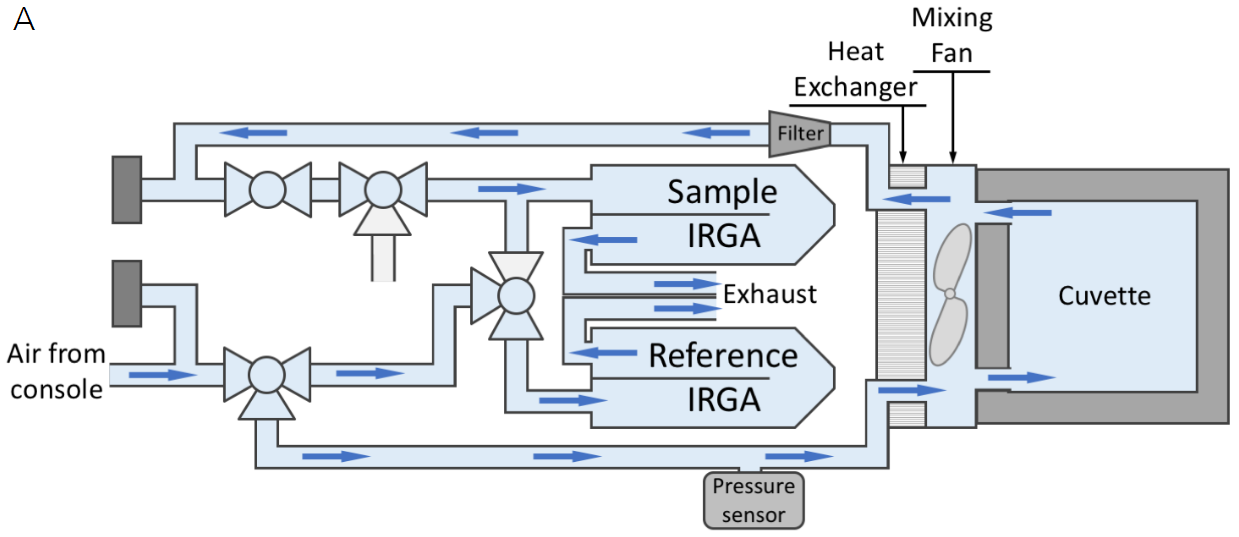
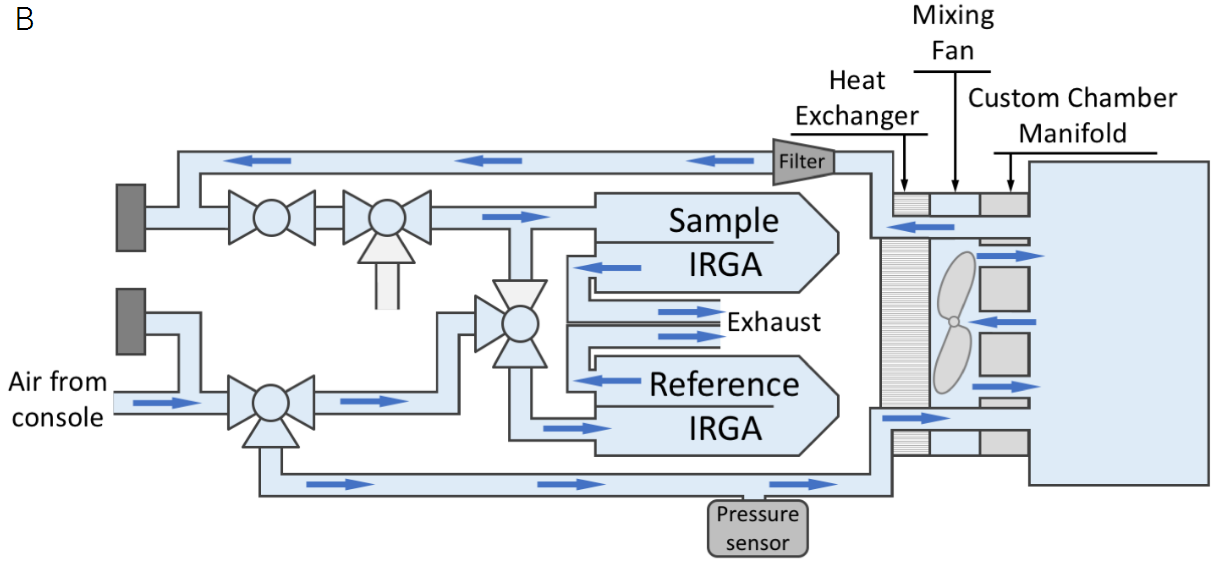
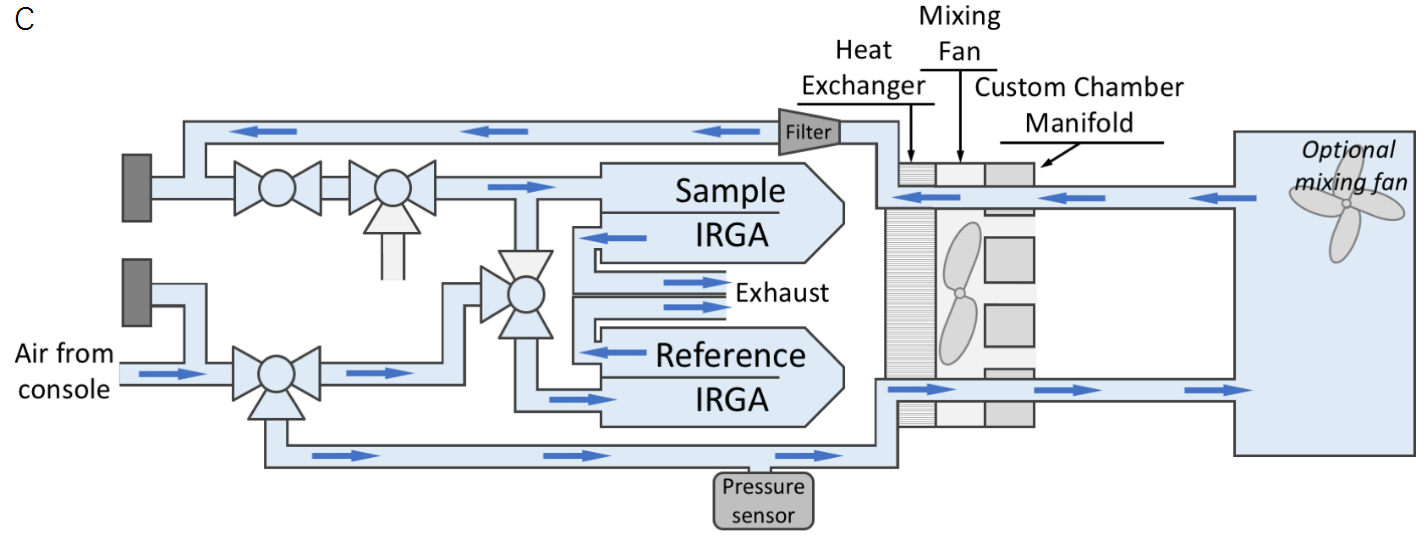
The flow path for the LI-6800 with our standard 3×3 chamber is shown in Figure 1‑1A. In typical operation, the LI-6800 is an open flow-through system, meaning that the air continuously flows through the cuvette and exhausts to the atmosphere. This flow path necessitates the use of two IRGAs, one to measure the incoming air stream (termed the reference) and another to measure the chamber (termed the sample) concentration. The custom chamber interface mounts to the sensor head similarly to the standard chambers and provides the user a couple of options for interfacing a custom chamber to the sensor head; the custom chamber can be mounted flush to the interface (flush-mount, Figure 1‑1B), or connected through tubing (tubed, Figure 1‑1C).
There are some important differences between a flush-mount and a tubed connection to the chamber. First, only a flush-mount chamber can take advantage of the built-in mixing fan to mix the chamber air. A tubed chamber would need to either be small enough to have well-mixed air relying only on the system air flow (e.g., the insect respiration kit, 6800-89) or would require installation of a user-provided mixing fan. The front of the custom chamber interface includes a 0-19 V power supply for this purpose if an additional mixing fan is needed. Second, only the flush-mount chamber can utilize the temperature control of the LI-6800 (note though that the range will be limited in larger chambers), as there would not be enough airflow to impact temperature in a tubed chamber.
Considerations for chamber design
Regardless of the method chosen to connect the chamber to the sensor head, several important considerations occur for design of the chamber itself. The volume of the chamber impacts several important operating parameters of the system which are discussed here.
Chamber volume and time constant
The total volume of a chamber should be carefully chosen to optimize measurements. The chamber obviously needs to be large enough to fit the desired material. A larger volume, however, will also take a longer time to flush, which means a longer time period to reach stable conditions and take a measurement. Designing the smallest chamber that will work for the material to be measured is advantageous to allow more rapid measurements. Larger surface area of chamber material can also lead to larger error due to water sorption and desorption from the chamber walls (see Chamber materials for a more thorough discussion). For a well-mixed open flow-through system, the chamber concentration is described by:
1‑1
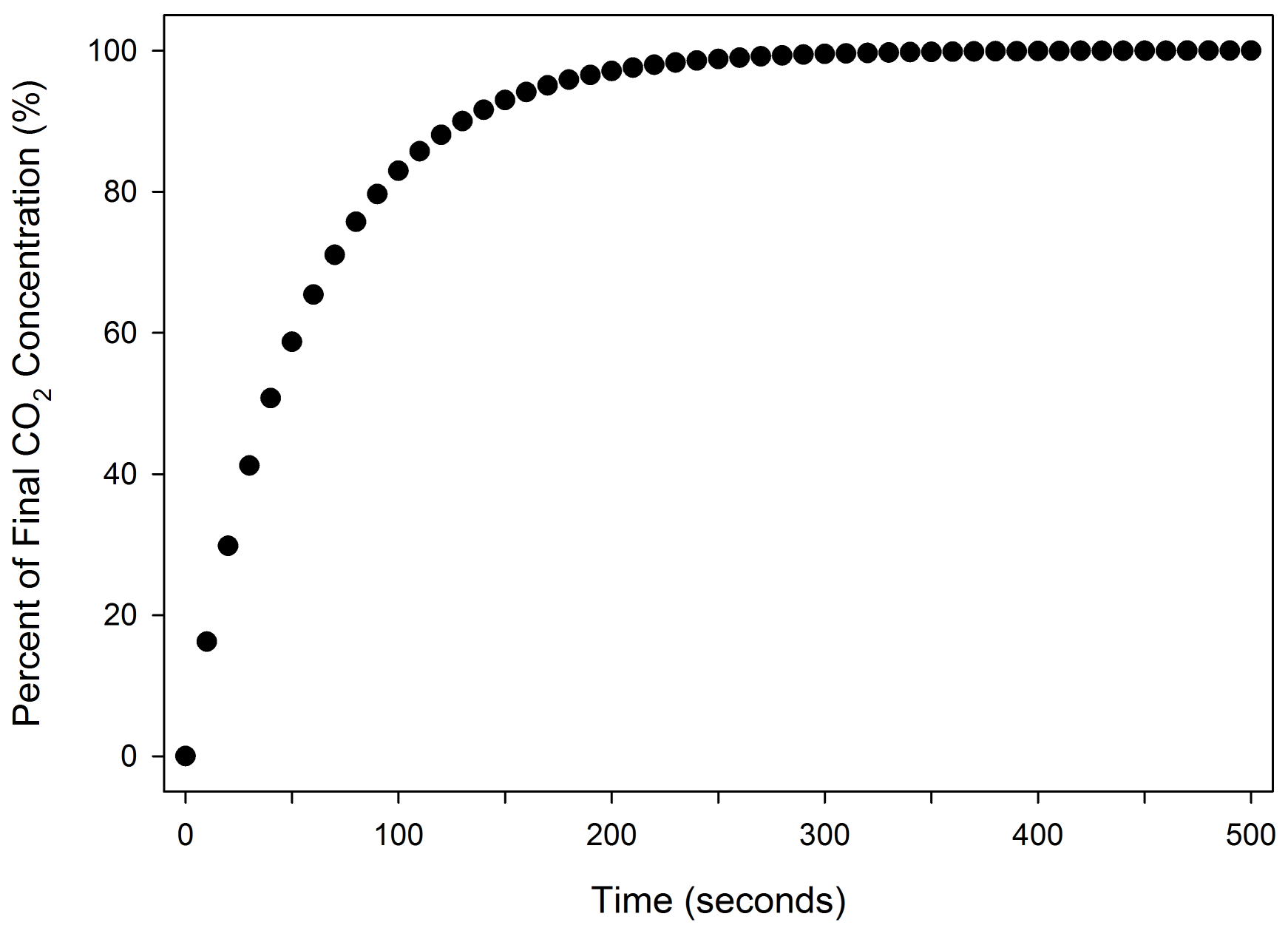
Where Co (µmol mol-1) is the initial chamber concentration, Ce (µmol mol-1) is the concentration entering the chamber, f (µmol s-1) is the flow rate, and V (cm3) is the chamber volume. The time constant τ of a chamber is, by definition, the time required to reach 1/e or ~63% of the final concentration. Flow and volume govern the τ of a given chamber:
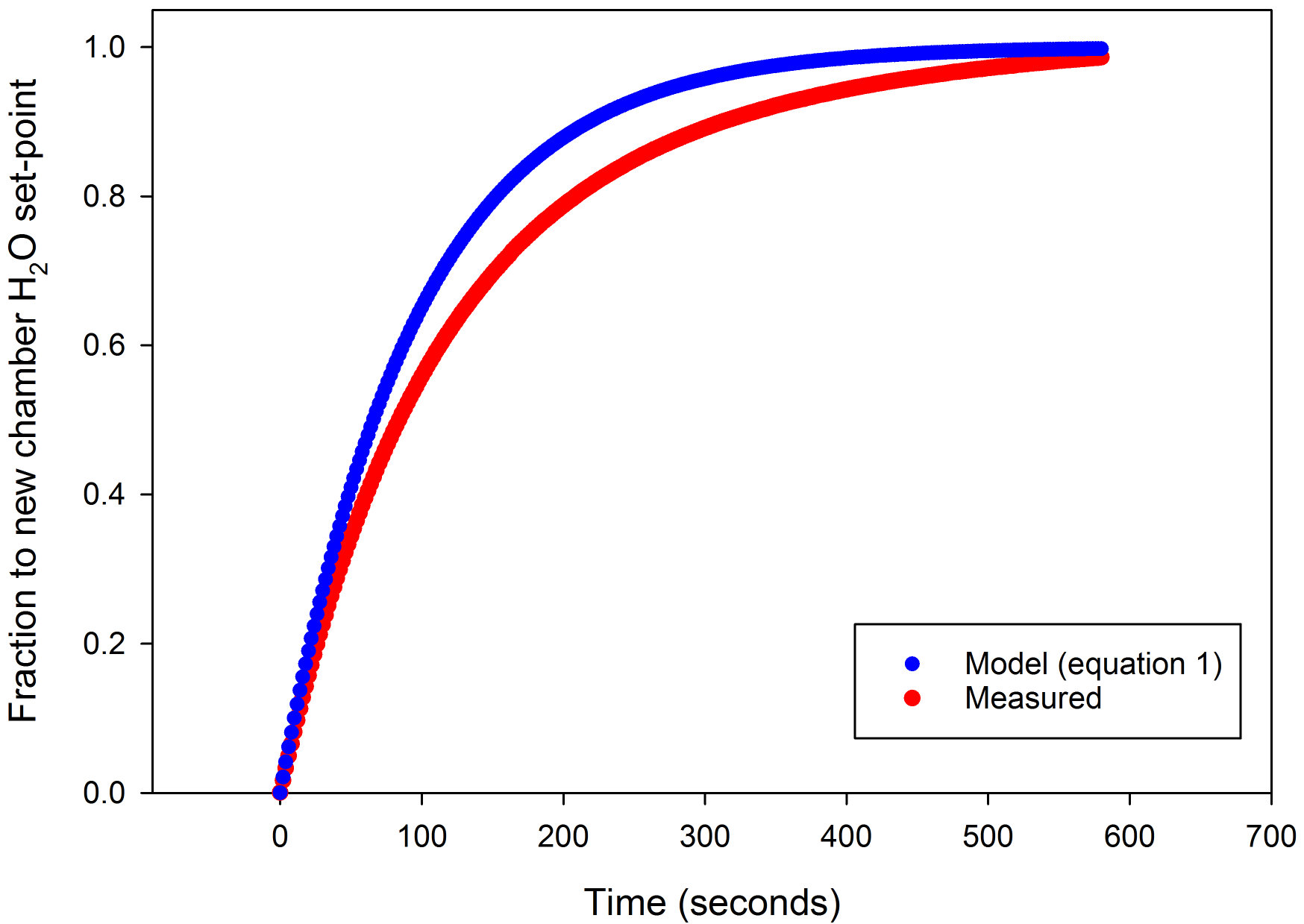
For example, in a 2 liter chamber with the maximum sample flow rate of 1400 µmol s-1 (35.4 cm3 s-1), the time constant is 56.5 seconds (Figure 1‑2). In contrast, the 6800-02 MPF fluorometer chamber has a volume of 83.6 cm3 and a time constant of 2.3 seconds at that flow rate. A 2 liter chamber would require 170 seconds to reach 95% of a change in concentration at that flow rate, meaning that when a change in the chamber is imposed it will take at least that long to reach stability. The volume of the chamber will have consequences on the speed at which measurements can be made.
Leaf area, flow rate and chamber humidity
Actively transpiring leaf material in a chamber will add water vapor to the chamber air, thus raising the humidity levels. Care must be taken to avoid condensation in the chamber, and especially condensed water into the optical path. The following analysis provides a guideline on chamber humidity as a function of the leaf area and expected conductance of the plant material. A chamber should be designed to prevent condensation under typical measurement conditions.
Conductance, g (mol m-2 s-1) can be expressed as
1‑3
Where f is the flow rate (µmol s-1), Wx is the water mole fraction (mmol mol-1) in the reference air (r), sample chamber (s) and leaf inter-cellular air space (l), respectively, and S is the leaf area (cm2). Conductance can be approximated as
Solving equation 1‑4 for Ws yields
1‑5
Where
Assuming the chamber air temperature and internal leaf temperatures are the same, condensation will occur when Ws approaches Wl. Figure 1‑3 shows the relationship between chamber water mole fraction and sample flow rate for several combinations of conductance and area. For example, at a flow rate of 800 µmol s-1 , a conductance of 0.16 mol m-2 s-1 and an area of 50 cm2, Ws would be approximately half-way between Wr and Wl. If leaf temperature were 30 °C, Wl would be ~43 mmol mol-1 (see the LI-6800 manual, equation A-43 and A-41 for computing water mole fraction from leaf temperature), so for dry incoming air (Wr = 0 mmol mol-1), Ws would be ~21. 5 mmol mol-1. If the temperature of the LI-6800 sample IRGA (Ts in the status group), was also 30 °C, then both chamber and IRGA relative humidity is ~50% and safe from condensation.
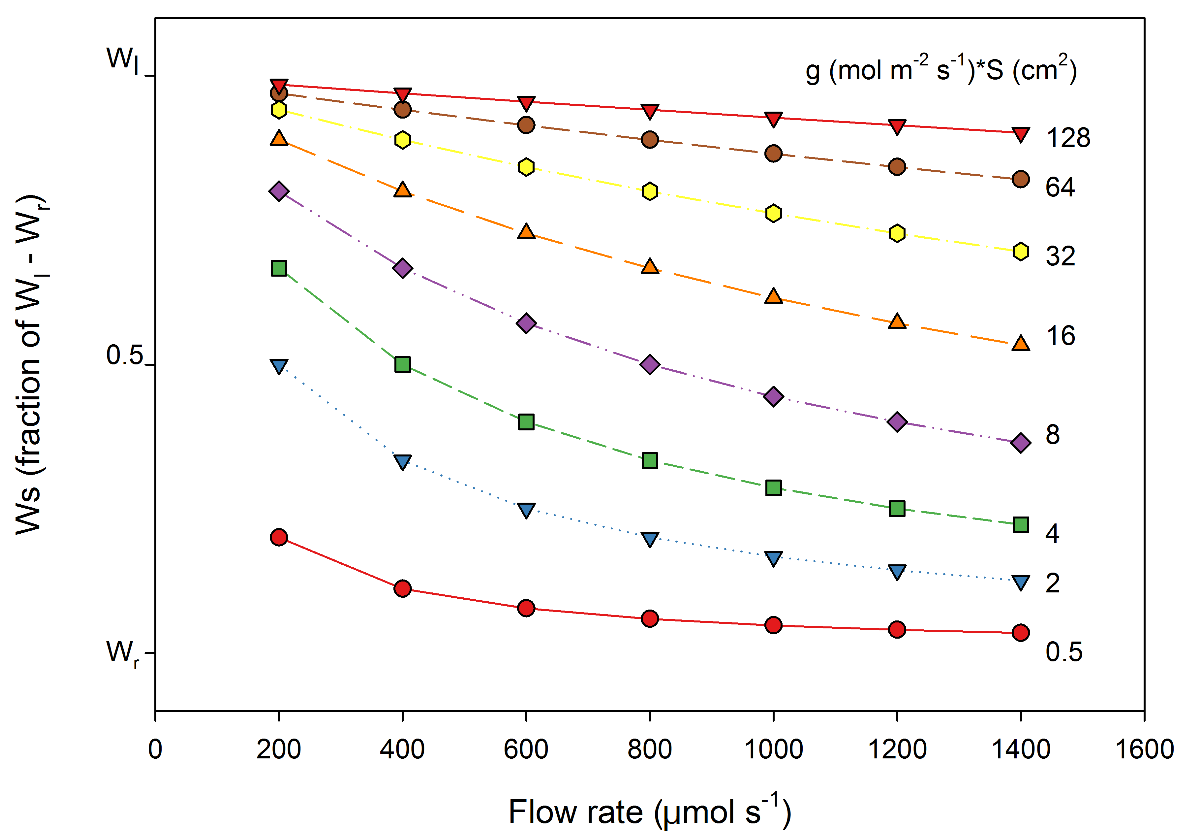
Leaf area, flow rate, and CO2 differentials
Measurements on plant (or animal) material with very low gas exchange rates may require the use of a larger custom chamber, as increasing the amount of material in the chamber can increase the precision of the measurement. The increased precision arises from larger differences in the concentrations measured by the sample and reference gas analyzers. The amount of material required is dependent upon the expected flux rates, the desired measurement precision, and the noise of the system. The following analysis, while not a complete error analysis on all inputs into the flux calculation, aids in determining the amount of material needed to obtain a reasonable amount of precision in the LI-6800.
The expected error in the measurement can be determined by:
where ΔCO2error is the maximum acceptable error in ΔCO2, ΔCO2 is the difference between the reference and sample gas analyzers, and NoiseCO2 is the total noise present in the measurement of CO2 concentrations.
Rearranging equation 1‑6:
1‑7
In order to determine the ΔCO2 required, the system noise must first be quantified. The system noise can vary between instruments and instrument settings, and should be measured directly on the instrument. To do this, set up the instrument to the desired flow rate and CO2 concentration and add CO2s and CO2r to the variables monitored in Stability Criteria. Set the stability criteria period to a desired time (we chose 60 seconds) and monitor the standard deviations shown in the stability screen.
The total noise on ΔCO2 can be calculated by converting the standard deviations to peak-to-peak noise for a given confidence internal. We will choose to use 3 standard deviations (which encompasses 99.7 % of the data points expected).
1‑8
Where SDx is the standard deviation measured by the instrument and x is the reference or sample analyzer. The actual concentration measurement at any given time will be less than ½ of the peak-to-peak noise for each analyzer. As ΔCO2 is the difference of the concentrations measured from two analyzers, the total noise in ΔCO2 is the sum of the noise of the two analyzers.
1‑9
In an instrument using default 4-second averaging, the standard deviation on both the sample and reference analyzers was measured at 0.02 µmol mol-1. From those measurements, the calculated NoiseCO2 is 0.06. If the desired measurement precision was 2%, then the required ΔCO2 to achieve that precision is 3 µmol mol-1.
The final step in the analysis is to determine how best to achieve the required ΔCO2. The ΔCO2 will be a function of the system flow rate, the amount of material in the chamber and the assimilation rate of the material. Ignoring the impacts of water vapor dilution in an open system, the assimilation rate can be computed as:
1‑10
Where A is the assimilation rate (µmol m-2 s-1), f is the flow rate (µmol s-1), and S is the leaf area (cm2). The equation can be rearranged to solve for S:
1‑11
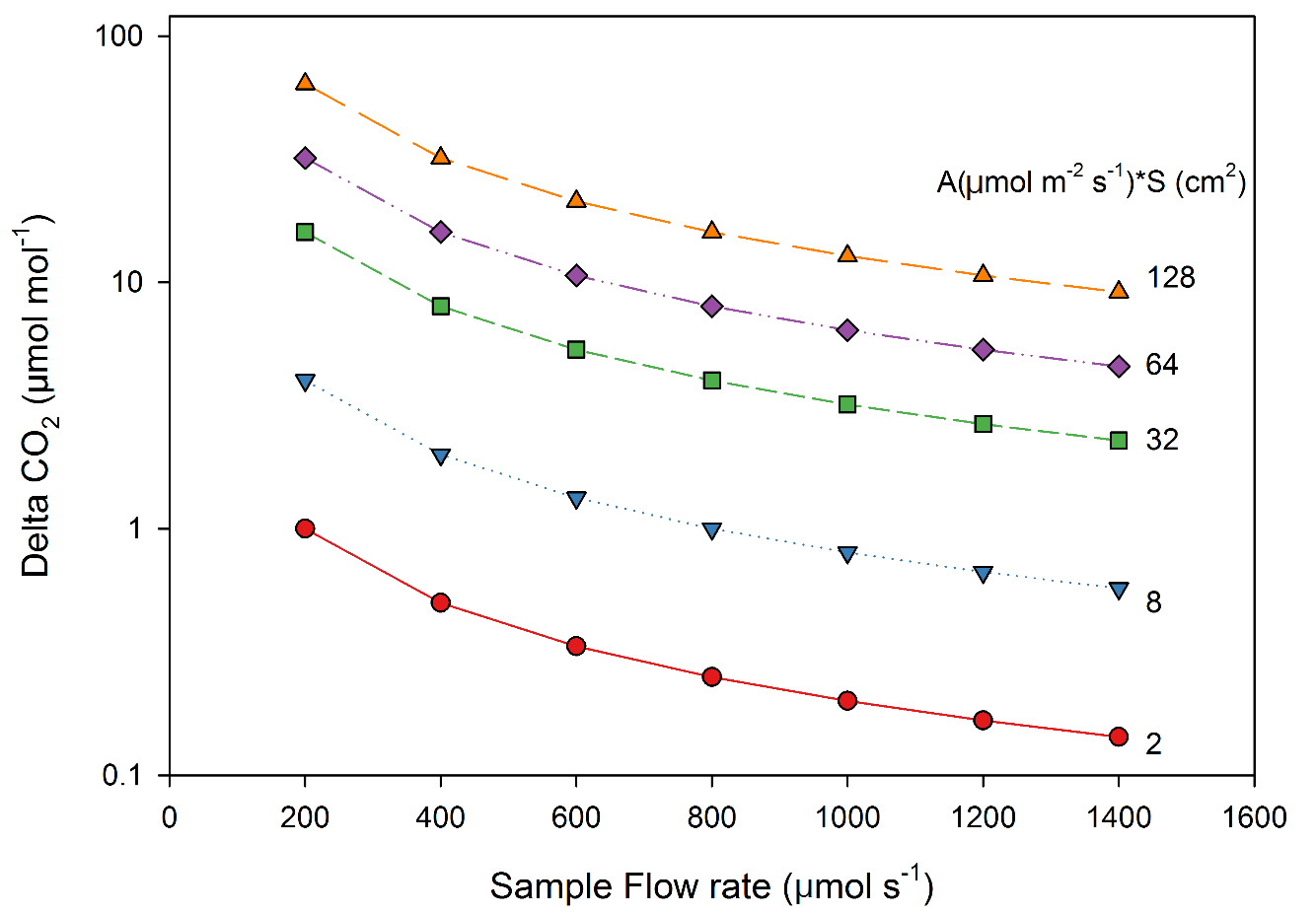
Now we can calculate the area of material needed to generate a given CO2 differential at a given assimilation rate. For example, for an assimilation rate of 0.5 µmol m-2 s-1, a leaf area of 20 cm2 is required to generate a 2% measurement precision (or 3 µmol mol-1 ΔCO2).
Figure 1‑4 shows ΔCO2 generated as a function of flow rate for multiple combinations of assimilation rate and leaf area. Note the strong dependence of this relationship to flow rate, not just to leaf area in the chamber. Consider this carefully before choosing to design a chamber solely for the purpose of measuring low assimilation rates, as optimizing measurements with the standard chamber may be just as effective; for example a leaf filling the standard 3×3 leaf chamber (9 cm2) with a low flux rate of ~0.9 µmol m-2 s-1, would generate a ΔCO2 of <1 µmol mol-1 at a flow rate of 1000 µmol s-1, but ~4 µmol mol-1 at 400 µmol s-1, which would generate very precise measurements based on the analysis above. Flow rates below 200 µmol s-1 are not recommended as lower flow rates may cause measurement noise through the diffusion into the analyzer exhaust ports.
Chamber mixing and temperature control
As noted above, only chambers that are flush-mounted to the custom chamber interface can utilize the LI-6800 sensor head mixing fan and temperature control. For chambers connected via tubing, air mixing will have to be supplied through an external mixing fan. Larger flush-mount chambers may also require additional mixing. We have tested a 2.5 L chamber and found adequate mixing with the internal mixing fan running at 14000 RPM. The range of temperature control will be a function of the chamber volume. For example, in a 2.5 liter chamber with no solar loading, the capacity to cool the chamber air temperature was reduced to ~6 °C below ambient. With this chamber, though, the instrument was still able to warm the air to 10 °C above ambient.
Chamber materials
The most important consideration for the material used to build custom chambers is the sorption/desorption of water vapor. On many surfaces, water vapor will slowly sorb and desorb, leading to long equilibration times, particularly in large chambers with lots of surface area. If water vapor does not reach equilibrium, there will be error in the recorded transpiration rates. Choosing a material with low rates of water sorption can reduce this impact. The material we have found with the best performance for water vapor sorption is nickel plated aluminum. For plastics, cell cast acrylic performs well. Raw aluminum and extruded acrylic products should be avoided due to strong interactions with water vapor.
An example using a 2.4 liter cell cast acrylic chamber (Figure 1‑5) demonstrates that water sorption does result in deviation from modeled wash-out times. Where large surface areas will be present in the chamber or less than optimal materials must be used, the chamber can be lined with Teflon tape, such as Propafilm, which can reduce the impact of water sorption.
Regarding chamber pressure
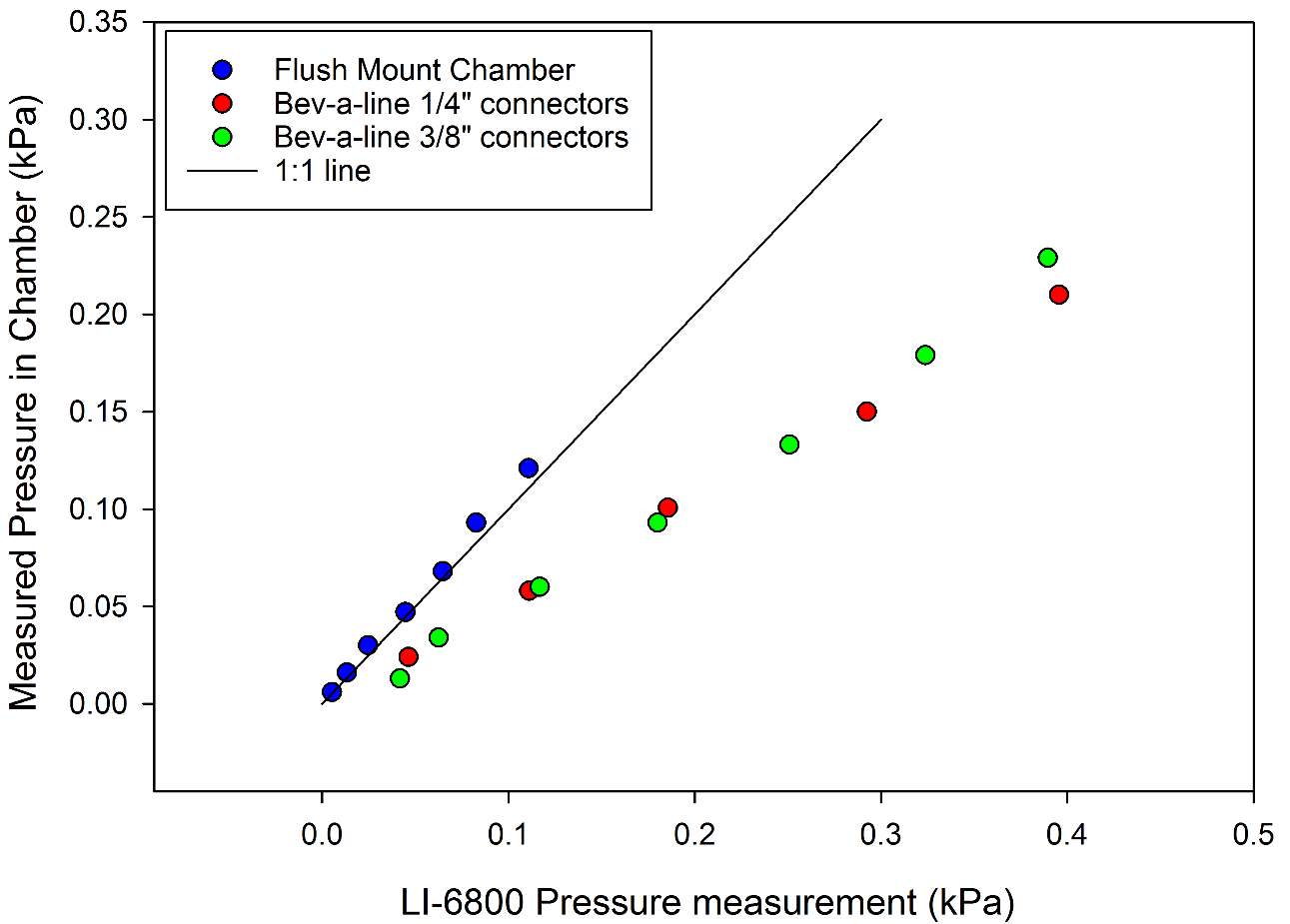
The LI-6800 includes automatic chamber pressure control, with a user-controllable valve located directly after the sample chamber (Figure 1‑1). While the user can increase chamber pressure, the minimum chamber pressure is a function of the chamber mounting and the sample flow rate (Figure 1‑6). For example, at a flow rate of 800 µmol s-1, a flush-mount chamber would have a native pressure of 5 Pa. Using the larger 3/8” connectors and 30 cm of Bev-a-line tubing with a tubed chamber produces ~9 Pa, and the smaller connectors 15 Pa. Connector size, tube diameter and tube length will all impact the native pressure in the chamber relative to the atmosphere.
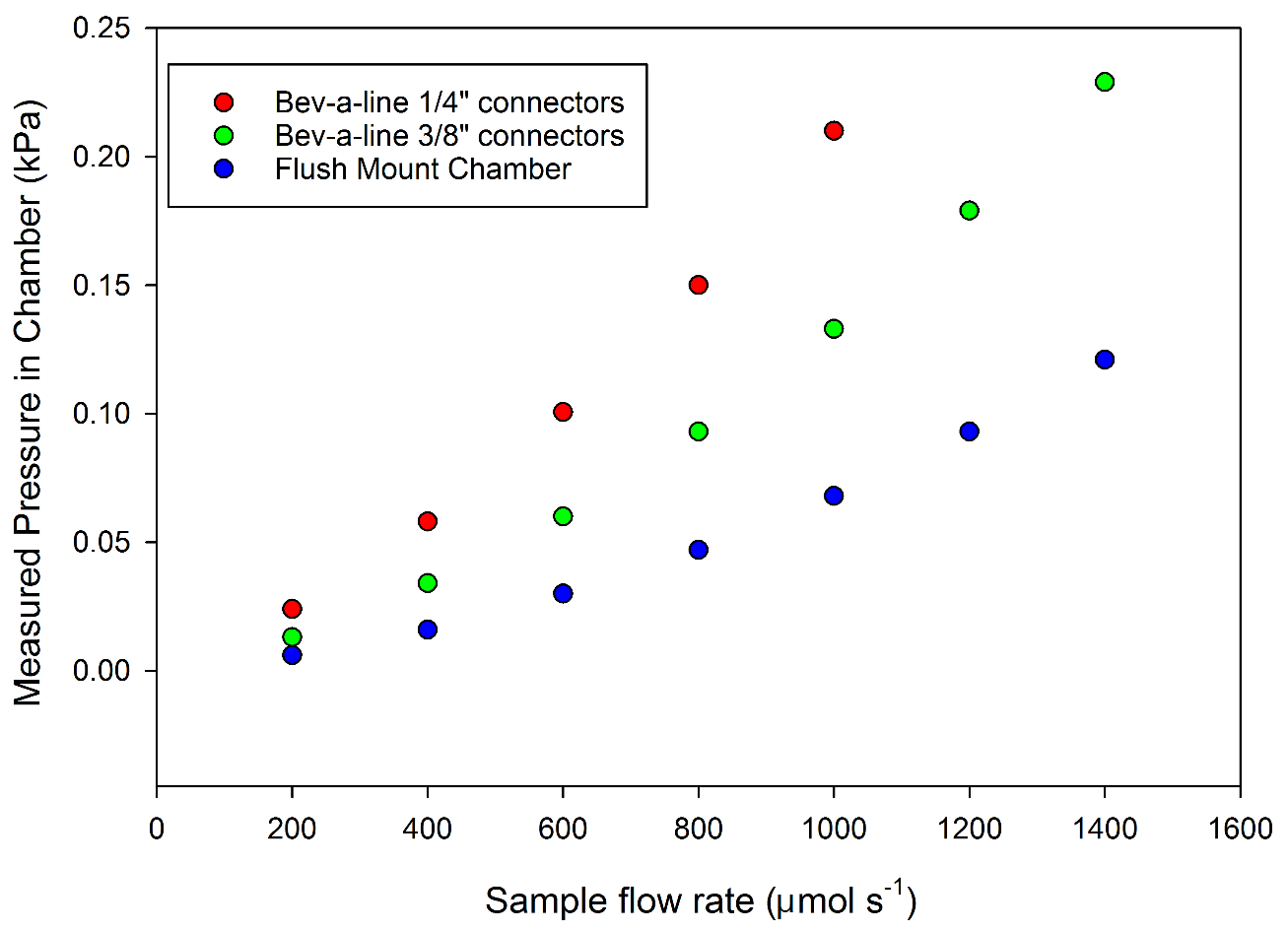
In Figure 1‑6, the chamber pressure was measured directly with an external pressure sensor. The LI-6800 sensor head contains a differential pressure sensor to monitor chamber pressure, depicted as Pressure Sensor in Figure 1‑1. The internal pressure sensor is located upstream of the chamber in the flow path. Due to the upstream location of the pressure sensor, the pressure measured by the LI-6800 sensor will have an offset from the actual chamber pressure (Figure 1‑7). With a flush-mount chamber, there is little-to-no pressure drop between the sensor and the chamber, and the pressure sensor provides an accurate chamber pressure. With a tube-mounted chamber, the chamber pressure will be less than that as measured by the internal pressure sensor. While for most measurement cases, this difference will be small, it may be important for some measurements.
Calculated flux units
An important consideration when computing fluxes is the metric by which to normalize the fluxes. The LI-6800 currently allows flux calculation on an area-basis or a mass-basis. This is chosen in the Start Up > Chamber Setup screen. Area-based fluxes will normalize to leaf area with fluxes in units of µmol m-2 s-1 (assimilation) and mol m-2 s-1 (transpiration) with the user entering area in cm2, as with standard leaf chambers. Mass-based fluxes will compute fluxes in units of µmol kg-1 s-1 (assimilation) and mol kg-1 s-1 (transpiration), with users entering mass in grams.
Chambers connected with tubing
This section discusses considerations for using tubing to connect a chamber. A common application for this type of chamber would be for respiration of insects or other non-plant material using a chamber like the 6800-89 Insect respiration chamber. Additionally, for samples that require very high humidity or even liquid water in the chamber, this configuration would be practical, as the tube returning from the chamber can be run through an ice bath to dry out the air to prevent condensation in the IRGA.
Connecting the chamber
Threaded adapters (compression to 7/16-20 straight thread) are provided in the custom chamber kit that can accommodate either ¼” OD or 3/8” OD tubing (only ¼” OD tubing is provided in the kit) to allow for tubed connections between the chamber and mounting plate. The compression side of the connectors comes with brass ferrules installed and additional nylon ferrules are provided in the kit. To make connections to plastic tubing, such as the Bev-a-line provided in the kit, it is recommended that the nylon ferrules be used.
In a tubed configuration, the mixing fan ports should be covered (plugs are included, part # 300-16271) to prevent dust/debris from entering the mixing fan area while the chamber is in use. The mixing fan should be turned off during measurements with this mounting method.
Following from the above discussion, when considering a chamber connected through tubing, the following items should be considered:
- Chamber mixing: The sensor head mixing fan cannot be used, so either the chamber should be small or an external mixing fan should be used and powered through the auxiliary power supply.
- Temperature control: The LI-6800 temperature control module is not available with this type of chamber mount.
- Chamber pressure: The actual chamber pressure will be a function of the length and diameter of the tubing
- System response time: Response time will be impacted in two ways. Along the tube we can assume flow, leading to a transport delay that is a function of flow, length and cross-section. In the chamber, there is a delay based on the chamber time constant (flow and volume from equation 1‑2).
Chambers flush-mount to interface
This section discusses considerations for chambers that are flush-mounted to the custom chamber interface. A common application would be for larger samples than cannot fit into standard chambers, or for measurements of short canopies such as turfgrass. Two examples are provided, showing measurement of banana respiration with respect to temperature and assimilation of grass as a function of ambient photosynthetically active radiation (PAR).
Connecting the chamber
A flush-mounted chamber must be built to the correct dimensions to screw directly onto the custom chamber interface; a 1:1 drawing is provided (). When looking at the interface from the chamber perspective, the incoming flow is on the upper left, outgoing flow is on the bottom right, the other corners are air from the mixing fan and the center hole is return to the mixing fan. If the internal mixing fan is to be used, all 5 holes are needed to connect to the chamber. There are 6 screw holes included on the plate to attach the chamber with and o-rings are included to seal the chamber. All necessary dimensions are provided in .
Following from the above discussion, when considering a flush-mount chamber, the following items should be considered:
- Chamber mixing: The internal mixing fan can be used with this chamber, but may not provide sufficient mixing for very large chambers. We have found adequate mixing up to ~2.5 liters
- Temperature control: Temperature control range will be a function of the chamber volume and ambient conditions, and will likely be reduced from our specification of ±10 °C.
- Chamber pressure: Chamber pressure will be properly measured with the internal pressure sensor. Note that for adequate flow to reach the sample IRGA the chamber must be relatively well sealed.
Example 1: Effects of temperature on fruit respiration
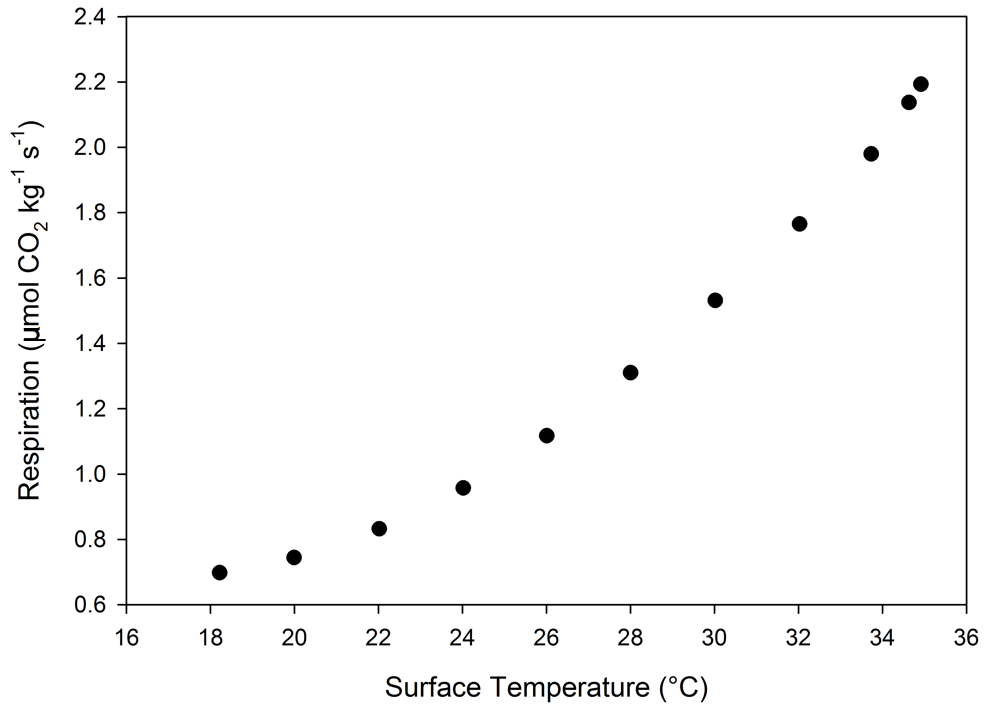
In this example, a 2.5 L flush-mount chamber was used to measure respiration from a banana (Figure 1‑8) that weighed ~185 grams. The chamber was sealed through a bottom plate of acrylic and closed cell foam weather stripping materials, which are available at most hardware stores. A poorly sealed chamber will result in no flow to the sample gas analyzer and measurements cannot be made. In this chamber, mixing was provided solely by the internal mixing fan run at high speed (14,000 RPM).

The respiration rates of the banana were measured as a function of the chamber temperature (Figure 1‑9). In this example, with a flow rate of 1400 µmol s-1, the ΔCO2 ranged from 100 – 300 µmol mol-1 over the range of temperatures measured. Despite the low flux rates (0.6 – 2.2 µmol kg-1 s-1), the ΔCO2 allowed for a high degree of measurement precision.
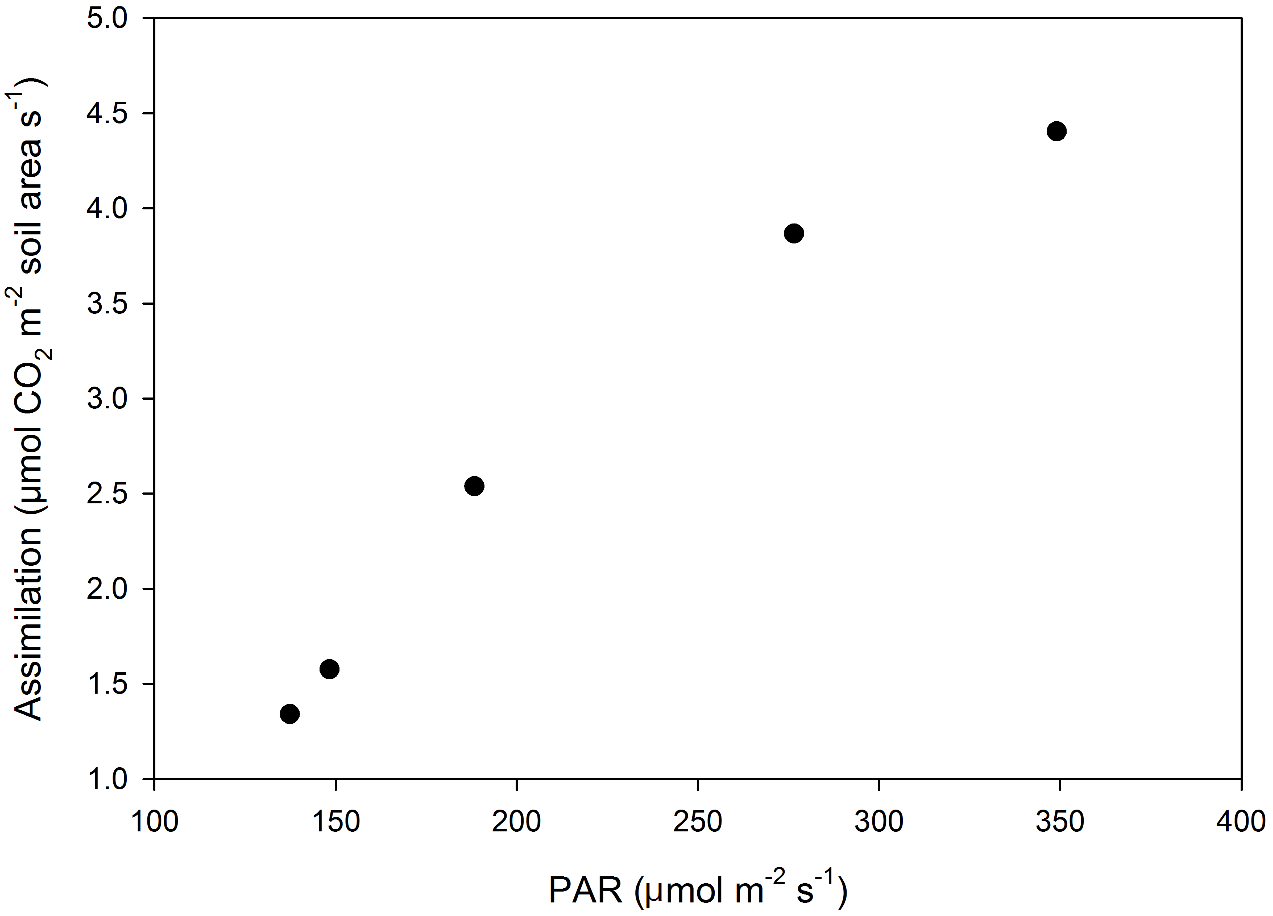
Example 2: Effects of PAR on turf assimilation
A larger chamber (~4 liters) was used to demonstrate measurement of turfgrass assimilation (Figure 1‑10). In this example, measurements were made using a PVC soil collar of 20 cm diameter, or a surface area of 317.8 cm2. The chamber was sealed to the area using closed cell foam weather stripping materials available at most hardware stores. It is important that the chamber seal properly as poor sealing will prevent air flow from the chamber to the IRGA and the chamber air will not be sampled.

In this large chamber, the internal mixing fan would not be sufficient to mix the air in the chamber. A second mixing fan was added and powered through the auxiliary power port on the sensor head to provide additional mixing. With a chamber of this size, the time constant or τ would be quite high, ~120 seconds. It will take many (~20) minutes for each measurement with such a large volume to flush. With the canopy height measured, a smaller chamber would have been sufficient and allowed for more rapid measurements.
Measurements were taken throughout the course of a morning and CO2 Assimilation plotted as a function of ambient PAR as measured using the LI-190R external quantum sensor on the LI-6800 sensor head (Figure 1‑11). Chamber soil surface area was 317.8 cm2 and reported fluxes are on a soil surface area basis. With such a large chamber volume, even at a maximum flow rate of 1400 µmol s-1, condensation could occur at higher light intensities with higher transpiration rates. Risk of condensation and the chamber time constant mentioned above are two reasons that a smaller volume chamber would be more appropriate for measuring short canopies such as turfgrass.
Conclusions
The LI-6800 custom chamber interface provides numerous ways to interface to a variety of custom chambers. Chambers in a variety of sizes and form factors are available from LI-COR. If none of the standard chambers meet the needs of your application, the custom chamber interface can extend the use of your LI-6800. There are several important design considerations discussed in this application note that should be considered before designing a custom chamber.
| Description | Part Number |
|---|---|
| Custom Chamber Kit | 6800-19 |
| Custom Chamber Adapter (1) | 9968-182 |
| Custom Chamber Spares Kit (1) | 9968-254 |
| Mounting Screws (6) | 150-14476 |
| Swagelok ¼" Fittings; 7/16 - 20; (2) | 300-16268 |
| Swagelok ¼" Vinyl Ferrule Set (2) | 300-05375 |
| Stainless Steel Plugs with O-ring (3) | 300-16271 |
| ¼" 90° Connectors; 5/16-24; (2) | 300-16515 |
| Bev-a-line tubing, ¼" (4.5 m) | 220-01824 |
| Swagelok ⅜" Fittings; 7/16 - 20; (2) | 300-16269 |
| Swagelok ⅜" Vinyl Ferrule Set (5) | 300-16272 |
| Auxiliary Power Cable (1) | 9968-260 |
| O-ring; 15×1.5 mm (2) | 192-16274 |
| O-ring; 10×1.5 mm (1) | 192-14435 |
| O-ring; 6×1.5 mm (1) | 192-14314 |
| Propafilm™ Kit | 9964-151 |
